No products in the cart.
With a solid foundation of research and clinical experiences, Gene Solutions’ R&D team has invented a new breakthrough technology called SPOT-MAS™ (Screen for the Presence Of Tumor by DNA Methylation and Size) that helps detect circulating tumor DNA (ctDNA), the biomarker for cancer at the early stages. With SPOT-MAS™, healthcare professionals are able to identify the presence of cancer even when patients do not have apparent syndromes or signals.

Cell-free DNA has been researched across the globe, especially in developed countries
Source: Illumina
Cell-free DNA is the non-unencapsulated DNA released into the blood stream, usually from the process of programmed cell death (as known as apoptosis) of both normal and abnormal cells. Current studies about cell-free DNA are conducted worldwide, mostly in developed countries, to address different types of diseases in human bodies.
Circulating tumor DNA (ctDNA) is a part of cell-free DNA, released from a tumor clone, reflecting exactly different features of the tumor. By analyzing ctDNA, we can understand more about the tumor and create a personalized treatment plan to make it more effective for each cancer patient. This is a remarkable solution for biotechnology research along with clinical practice. Usually, cell-free DNA technology is developed and applied to prenatal care, early cancer detection and screening, and cancer recurrence monitoring.
Starting in 2017, the founding team with 3 scientists from Gene Solutions are passionate for improving healthcare standards in Vietnam and other developing countries, utilizing cell-free DNA technology.
And 3 years later, in 2020, Gene Solutions successfully built up an ecosystem with a total of 28 genetic testing products to help with the prognosis of disease and support treatment in prenatal care and cancer detection and inherited chronic disease screening. In the obstetrics – pediatrics segment, Gene Solutions takes 80% of the market share in terms of non-invasive prenatal testing, helping thousands of pregnant women access genetic technology at ease and at more reasonable price.
Along with that, the applications of machine learning models and big data from the dataset of genomes from 350,000 genetic tests conducted in total, which help detect abnormal cases, increase diagnostic accuracy, and reduce the number of false positives. False positives cause a great deal of waste in diagnosis and make test takers worry about their own medical condition.
Outstanding achievements of Gene Solutions’ cell-free DNA technology
Offering a more simple, accurate and safer screening methods, Gene Solutions set a great milestone for genetic research and development in Vietnam.
- More than 70,000 triSure noninvasive prenatal tests have been conducted in Vietnam, the world’s first NIPT test capable of detecting anomaly and reducing false positives.
- Over 3,000 OncoGS Cancer Liquid Biopsy tests are conducted, the world’s first liquid biopsy technology to detect DNA-based gene fusion (ALK, ROS1).
- Working towards cancer early detection to identify the presence of circulating tumor DNA.
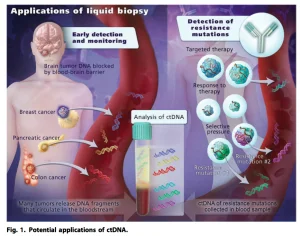
Cell-free DNA released from the tumor – the biomarker of cancer
Souce: enseqlopedia.com
With the outstanding advantage from the advanced machine learning system and large genetic data, the ECD solution has been able to do something those previous methods could not do, such as analyzing 4 unique markers from ctDNA at the same time combines genetic data from 20,000 cancer patients to detect cancer early in its asymptomatic stages. SPOT-MAS™ technology has a particularly important meaning in the prevention and detection of inherited factors causing cancer. The method accurately detects abnormal genomic factors of multiple common types of cancer including liver cancer, lung cancer, breast cancer and colorectal cancer, stomach cancer. Researchers now can conduct early cancer screening procedures with lower cost and fewer fault positives.

SPOT-MAS™ has been evaluated in K-DETEK™ program with the participation of 14 hospitals at top levels in Vietnam.

The community program “Towards a genetically healthy Vietnamese generation” aims to raise awareness about screening and early detection of cancer by donating 7000 free SPOT-MASTM cancer screenings, with a total value of more than VND50 billion, to more than 100 businesses, hospitals and clinics across the country.
» View detail «
Copyright © 2020 GENE SOLUTIONS
Legal Representative: Nguyễn Hữu Nguyên
Enterprise No. 0314215140 – HCMC D.P.I issued on January 23, 2017
Tư vấn di truyền